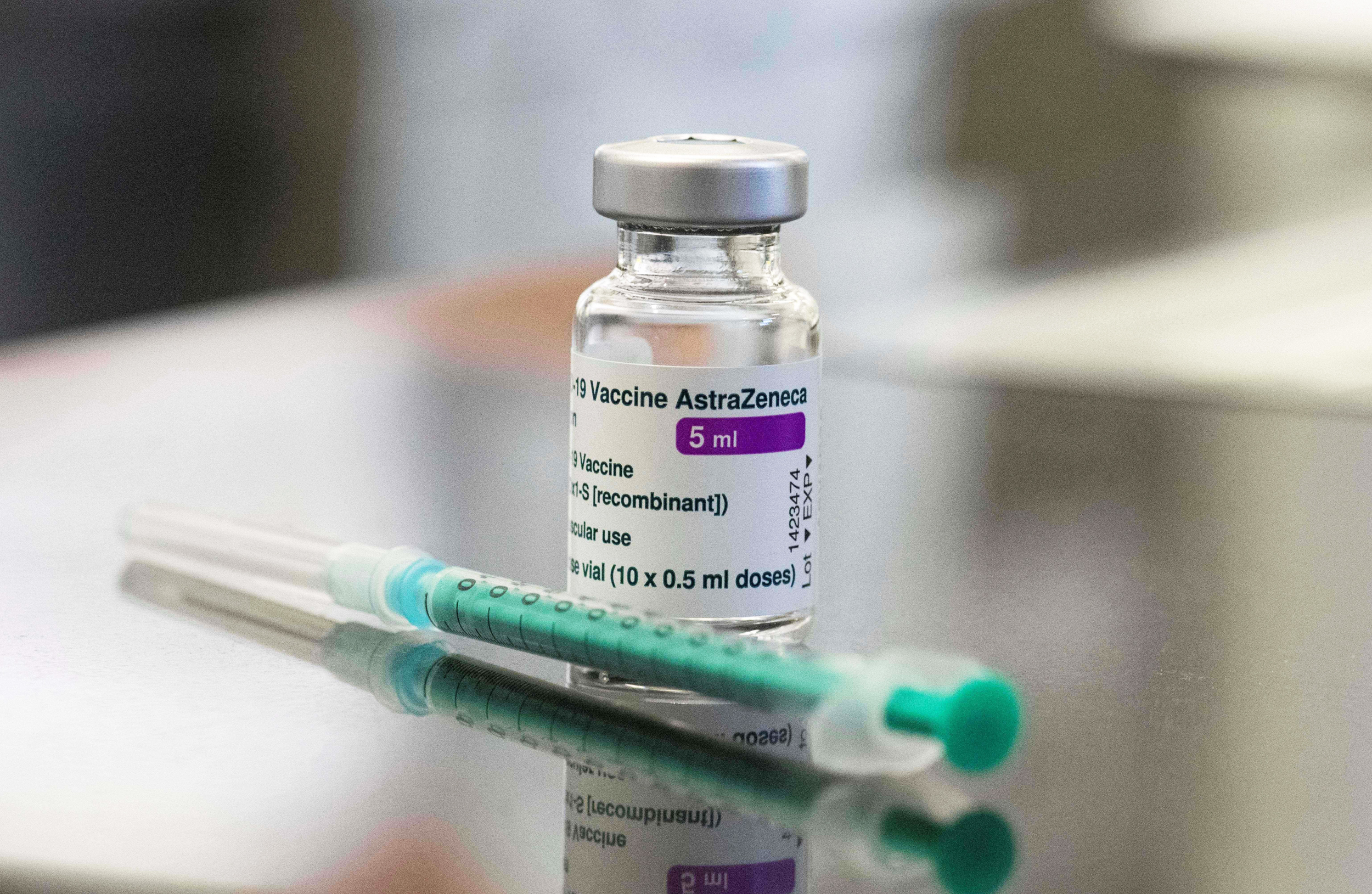
Germany and France are the latest countries to put their rollouts of the Oxford-AstraZeneca Covid vaccine on hold because of fears over blood clotting. Boris Johnson, Britain’s medicines regulator and AstraZeneca itself have all defended the vaccine as safe to use. Sky News has the story on Germany’s suspension.
Germany is suspending use of the Oxford-AstraZeneca coronavirus vaccine in order to investigate reports of blood clots.
The country’s Health Ministry said the measure was a “precaution”. …
In a statement, the German health ministry said the reported blood clots involved cerebral veins, but did not specify where or when the incidents happened.
It said its decision to suspend the vaccine was taken on the advice of national regulator, the Paul Ehrlich Institute.
The ministry said the EMA would decide “whether and how the new information will affect the authorisation of the vaccine”.
The Telegraph has the following on France:
Emmanuel Macron said that French authorities have decided to suspend shots at least until Tuesday afternoon, when the European Medicines Agency will issue its recommendation over the vaccine.
Yesterday, Ireland and the Netherlands followed the example of other EU countries – including Norway and Denmark – in halting their AstraZeneca vaccine rollouts amid reports of “bleeding, blood clots and a low count of blood platelets” in health workers who had recently received the vaccine. One particular batch of AstraZeneca vaccines (which is implicated in reports of a death) was sent to 17 countries.
The greater the concern over the vaccine, the greater the defence from others. On March 14th, AstraZeneca addressed safety concerns in a statement.
A careful review of all available safety data of more than 17 million people vaccinated in the European Union and UK with Covid Vaccine AstraZeneca has shown no evidence of an increased risk of pulmonary embolism, deep vein thrombosis or thrombocytopenia, in any defined age group, gender, batch or in any particular country.
So far across the EU and UK, there have been 15 events of DVT and 22 events of pulmonary embolism reported among those given the vaccine, based on the number of cases the Company has received as of 8 March. This is much lower than would be expected to occur naturally in a general population of this size and is similar across other licensed COVID-19 vaccines. …
Furthermore, in clinical trials, even though the number of thrombotic events was small, these were lower in the vaccinated group. There has also been no evidence of increased bleeding in over 60,000 participants enrolled.
Responding to Germany’s suspension of the jab’s rollout, Boris Johnson said the vaccine is “both safe and effective”:
“[The UK has] one of the toughest and most experienced regulators in the world.
“They see no reason at all to discontinue the vaccination programme… they believe that they are effective, highly effective in driving down not just hospitalisations but also serious disease and mortality. We continue to be very confident about the programme.”
Representatives of Britain’s Medicines and Healthcare products Regulatory Agency added that the evidence “does not suggest” the jab causes clots.
We are closely reviewing reports but given the large number of doses administered, and the frequency at which blood clots can occur naturally, the evidence available does not suggest the vaccine is the cause.
Worth reading in full.
Stop Press: Professor Anthony Harden, the Deputy Chair of the Joint Committee on Vaccination and Immunisation (JCVI), has hit back at claims that the vaccine increases risk of blood clotting, saying: “We’ve given 11 million doses here and there’s no evidence of increased risk of blood clots.” The JCVI can now be added to the list of organisations that have dismissed concerns about the AstraZeneca vaccine which includes the World Health Organisation (WHO), the European Medicines Agency (EMA) and the Medicines and Healthcare products Regulatory Agency (MHRA).












To join in with the discussion please make a donation to The Daily Sceptic.
Profanity and abuse will be removed and may lead to a permanent ban.
Michael Ball and Alex Jones on The One Show shortly to tell us not to worry, so that’s a relief.
The hypocrisy of these people! Its safe they cry without any trials of the ‘double blind’ type they insist that Ivermectin or HCQ must have before use. Somehow their use of the vaccine ‘in public’ is worth more than the millions using ivermectin/HCQ.
Have a look at who sits on the Board of the MHRA, its like the poacher turned gamekeeper. The likes of AZ regulate themselves.
https://www.gov.uk/government/organisations/medicines-and-healthcare-products-regulatory-agency/about/our-governance
All that was requested was simple answers to relatively simple questions; Mr regulator, have you got evidence from trials that these things don’t cause clots? Answer, silence to the specific questions just loads of noise and dissembling.
These blood clots may be a red herring, but they are not the only negative events being temporally associated with the injections. The “low count of blood platelets”, briefly mentioned, and particularly its rapid onset following injection, may be a compellingly distinctive phenomenon.