It’s well-known that flu-like illnesses are seasonal, and COVID-19 seems no different.
What is not well understood is what drives the seasonality. The flu season is in the winter months so the obvious candidate is temperature. However, closer analyses have tended to rule out a simple role for temperature, not least because seasonality occurs in places with very different climates.
Other potential candidates are sunlight (including UV radiation) and humidity. None is completely convincing, though sunlight has been shown to have a direct role in stimulating the immune system.
Now there’s a new kid on the block: pollen.
Martijn Hoogeveen is an independent researcher who has been exploring the role of pollen in driving seasonality. Pollen is known to be triggered or inhibited by meteorological factors. Hoogeveen explains:
Meteorological variables, such as increased solar radiation and temperature – among others the absence of frost – not only trigger flowering and pollen maturation, they also affect the pollen bio-aerosol formation: dry and warm conditions stimulate pollen to become airborne. Rain, in contrast, makes pollen less airborne, and cools the bio-aerosol down. Very high humidity levels (RH 98%) are even detrimental to pollen (Guarnieri et al., 2006).
The main idea is that the pollen plays a further, significant role in stimulating the immune system (such as when it causes hay fever), which has a strongly inhibiting effect on flu-like viruses. A further, more speculative idea is that “anti-viral phytochemicals in pollen” could directly inhibit viral aerosols in the air.
Hoogeveen illustrates the relationships.
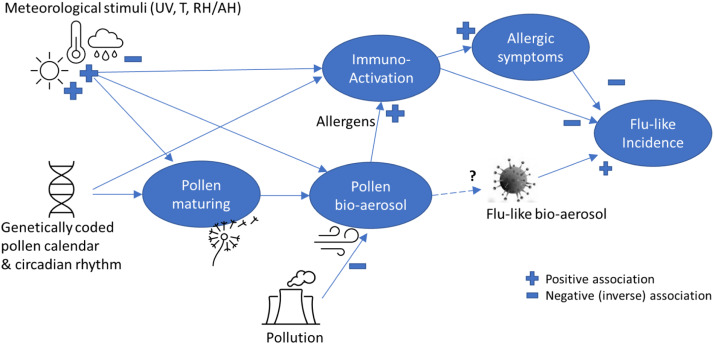
Hoogeveen observes that temperature by itself is not a good predictor of the flu season in the Netherlands.
The onset of the flu season, from mid-August in the Netherlands, coincides with an annual peak in hot, sunny days and is still in the middle of the summer season. … Although temperature strongly correlates with flu-like incidence (r(226) = −0.82, p < 0.0001), it has a negligible effect on ΔILI, weekly changes in flu-like incidence (r(224) = −0.02 n.s.), also when corrected for incubation time. Therefore, it seems unlikely that temperature has a direct effect on aerosol flu-like viruses and the life cycle of a flu-like epidemic. In line with this, temperature is also not a good marker for the onset or the end of the flu season. In the Netherlands the end of the flu season (Ro < 1) can coincide with an average temperature that is close to 0 °C and the start of the flu season (Ro > 1) can coincide with temperatures as high as 17 °C.
However, solar radiation and pollen, both of which are known to have an impact on immune activation, have a much stronger relationship with flu-like illnesses.
The following graph shows some of Hoogeveen and his team’s results, depicting the remarkable inverse correlation between flu-like incidence and pollen concentration.
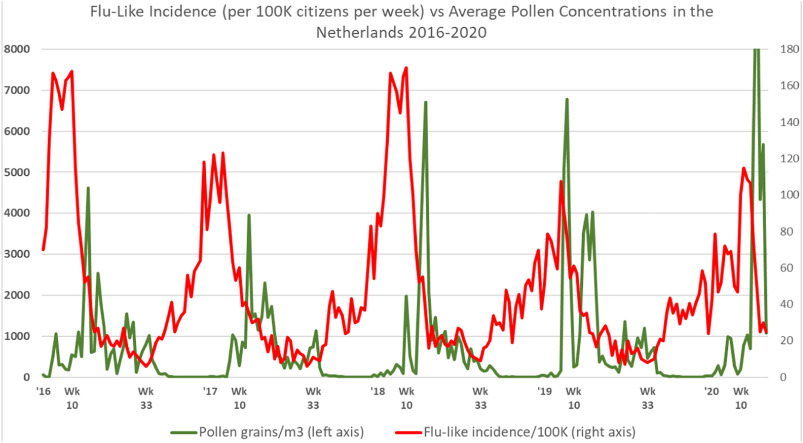
Hoogeveen explains:
Flu-like incidence starts to decline after the first pollen bursts. Moreover, flu-like incidence starts to increase sharply after pollen concentrations become very low or close to zero. This is a qualitative indication of temporality. Furthermore, we can notice that the first COVID-19 cycle behaved according to pollen-flu seasonality, at least does not break with it.
In formal terms: “The correlation for total pollen and flu-like incidence is highly significant when taking into account incubation time: r(222) = −0.40, p < 0.001.”
Hoogeveen identifies some thresholds that appear to trigger (or at least signify) the beginning and end of flu seasons.
We found that our predictive model has the highest inverse correlation with changes in flu-like incidence of r(222) = −0.48 (p < 0.001) when thresholds of 610 total pollen grains/m3, 120 allergenic pollen grains/m3, and a solar radiation of 510 J/cm2 are passed. The passing of at least the pollen thresholds preludes the beginning and end of flu-like seasons.
In the years covered in the study (2016-2020), Hoogeveen observed that “the pollen thresholds are passed in week 10 (± 5 weeks), depending on meteorological conditions controlling the pollen calendar and coinciding with reaching flu-like peaks, and again in week 33 (± 2 weeks), marking the start of the new flu-like season”.
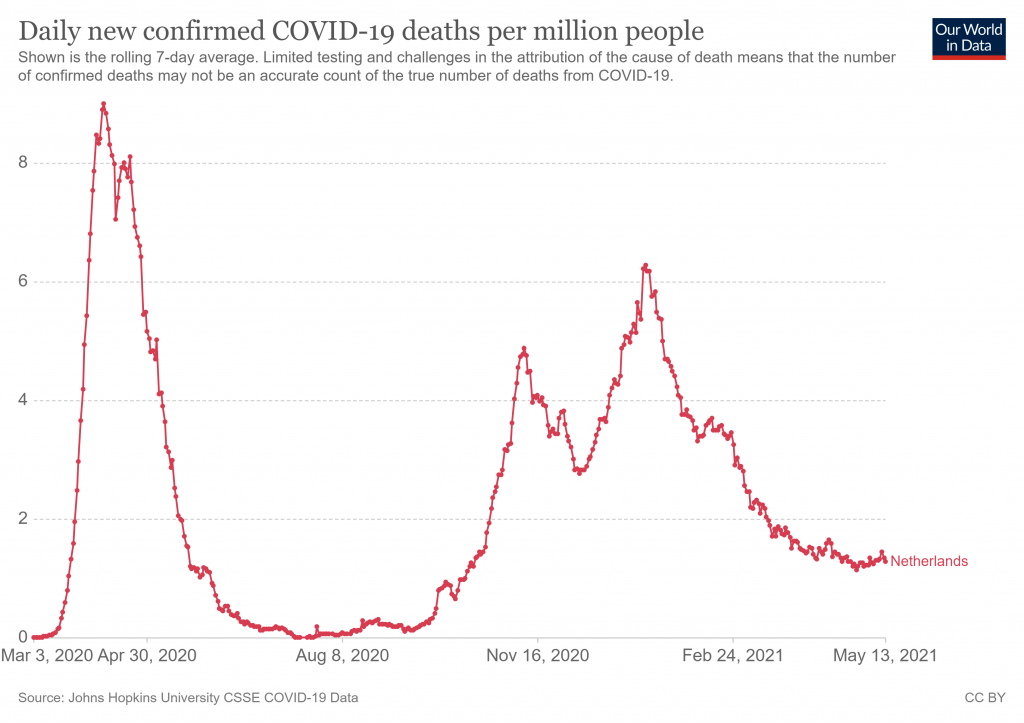
Hoogeveen doesn’t indicate how this model has fared in 2021 so far, though week 10 or even week five doesn’t appear to line up with the Covid peak in the Netherlands this winter (see below). It would be interesting to know the pollen concentrations over this time, and Hoogeveen’s thoughts about this.
Sceptics, who frequently point out that epidemics of Covid, just like those of other flu-like illnesses, are seasonal and surge and decline independently of policy interventions, will welcome research into the nature of this seasonality.
One unanswered question is the role of acquired immunity in triggering the peak and decline. If seasonality is the dominant factor then a peak may not have any relationship to the current level of population immunity acquired through infection and the fact that a wave has come and gone doesn’t tell us anything about how close we are to a protective level of population immunity. On the other hand, if seasonality interacts with population immunity then a peak and decline may tell us something about how far the population is immune.
Looking at the graph above, it seems to me that for 2016-17 in particular the peak (though not the decline) occurs well before there is any signal in the pollen. This suggests there is something besides pollen contributing to ending the exponential spread of the virus that year. Population immunity is one obvious candidate for this.
It’s also worth noting that in this model pollen and other drivers of seasonality are understood to operate primarily through their effect on the immune system. In this sense then the interaction with population immunity is built in.











Profanity and abuse will be removed and may lead to a permanent ban.